Joint Research Group Macromolecular Crystallography
Structure of the month - March 2010
Angew. Chem. Int. Ed. Vol. 48, 9954-9957 (2009), N. Klein et al., Copyright Wiley-VCH Verlag GmbH & Co. KGaA. Reproduced with permission.
A mesoporous Metal-Organic Framework: DUT-6
Nicole Klein#, Irena Senkovska#, Kristina Gedrich#, Ulrich Stoeck#, Antje Henschel#, Uwe Mueller*, and Stefan Kaskel#$
# Department of Inorganic, Dresden University of Technology, Mommsenstr. 6, D-01069 Dresden, Germany
* Helmholtz-Zentrum Berlin für Materialien und Energie, Soft matter and functional materials F-I2, BESSY-MX group, Albert-Einsteinstr. 115, D-12489 Berlin, Germany
$stefan.kaskel@chemie.tu-dresden.de,
www.chm.tu-dresden.de/ac1/
Abstract
Since the mid-90s Metal-Organic Frameworks (MOFs) are a novel class of porous materials attracting interest as adsorbents, catalysts and materials for separation processes. MOFs are formed by coordinative bonds of multidentate organic ligands to metal atoms or metal clusters. Resulting pores or channels are occupied by guest molecules (typically solvent used during the synthesis). The resulting structures are usually rigid and have large specific surface area and pore volume after solvent removal.
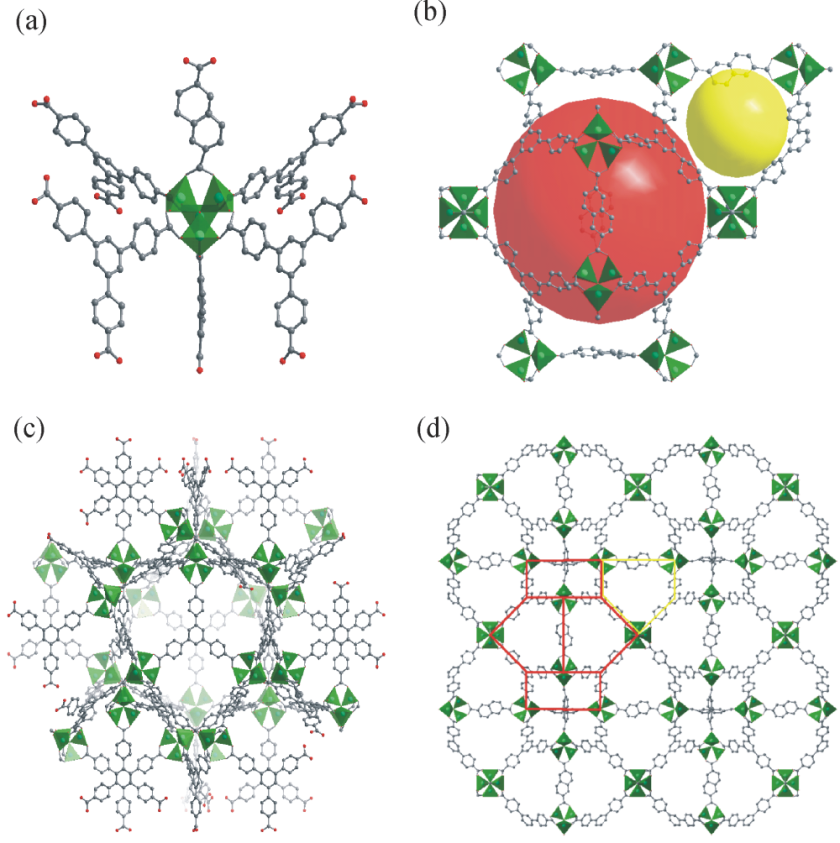
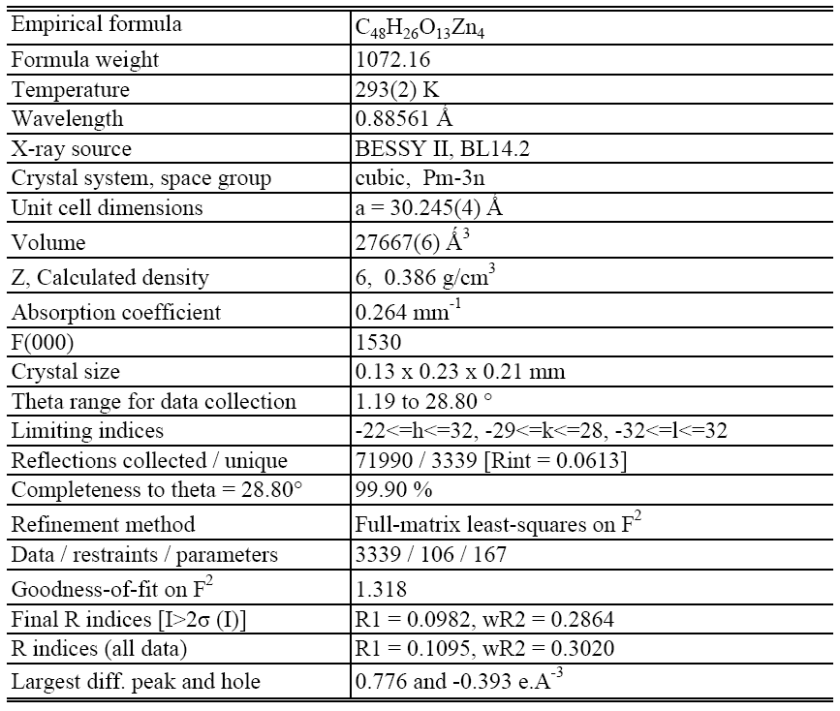
Six bridging carboxylate groups form an octahedral secondary building unit (SBU) with four btb linkers in a square arrangement while two additional 2,6-ndc linkers occupy the residual octahedral sites and further crosslink the network.Wide open dodecahedral mesoporous cages 2.5-3 nm in diameter are formed by twelve Zn4O6+ nodes, six 2,6-ndc linkers and eight btb linkers. A smaller second kind of pore consisting of four Zn4O6+ nodes, two 2,6-ndc linkers and four btb linkers are arranged to connect the larger dodecahedral pockets in order to form a periodic space filling. The wide open pores lead to an enormously high methane storage capacity (230 mg g-1, 100 bar) and to one of the highest hydrogen uptakes (60 mg g-1, 50 bar) that have been reported for MOFs so far.
DUT-6 is the synonym for: DUT = Dresden University of Technology; Zn4O(2,6-ndc)(btb)4/3, 2,6-ndc = 2,6-naphthalenedicarboxylate, btb = 1,3,5-benzentribenzoate.
This work was supported by the BMWT (FKZ: 0327796B).