Joint Research Group Macromolecular Crystallography
Structure of the month - July 2006
Embo Rep. Vol. 6, 2005, Pages 787-793
Structure of the TRAPP subunit TPC6 suggests a model for a TRAPP subcomplex
Daniel Kümmel1,2, Jürgen J. Müller1, Yvette Roske1,3, Rolf Misselwitz1, Konrad Büssow3,4, and Udo Heinemann1,2,*
1 Max-Delbrück Center for Molecular Medicine, Robert-Rössle-Straße 10, D-13092 Berlin, Germany
2 Chemistry Institute, Free University, Takustraße 6, D-14195 Berlin, Germany
3 Protein Structure Factory, Heubnerweg 6, D-14059 Berlin, Germany
4 Max Planck Institute for Molecular Genetics, Ihnestraße 73, D-14195 Berlin, Germany
*Correspondence: heinemann@mdc-berlin.de
Udo Heinemann
Max-Delbrück-Centrum für Molekulare Medizin
Robert-Rössle-Straße 10
D-13092 Berlin, Germany
Phone: +49 (30) 9406 3420
Fax: +49 (30) 9406 2548
Abstract
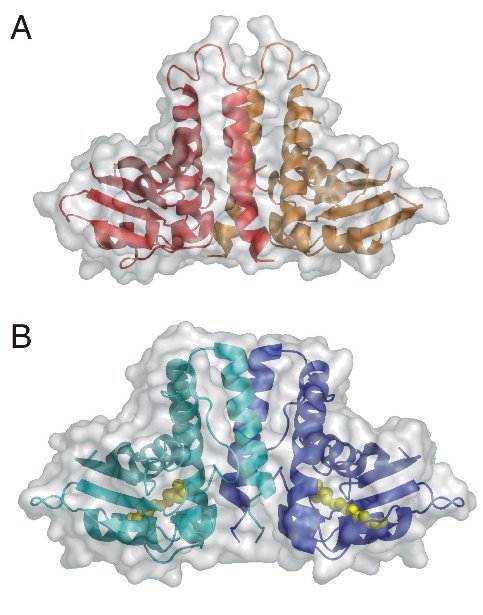
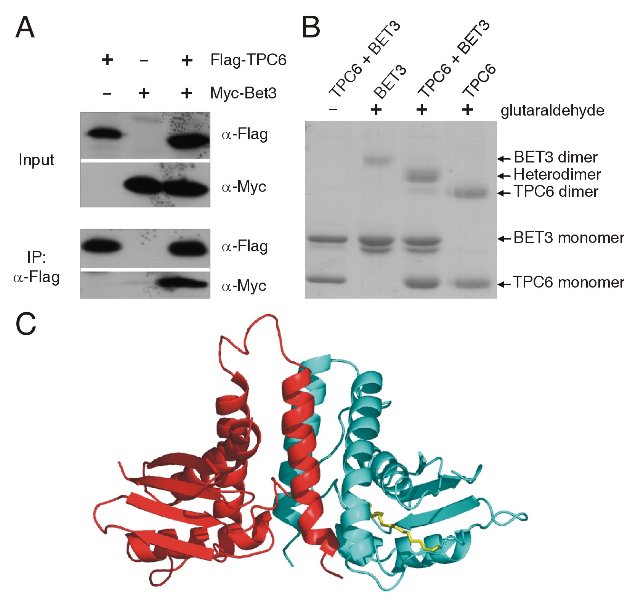
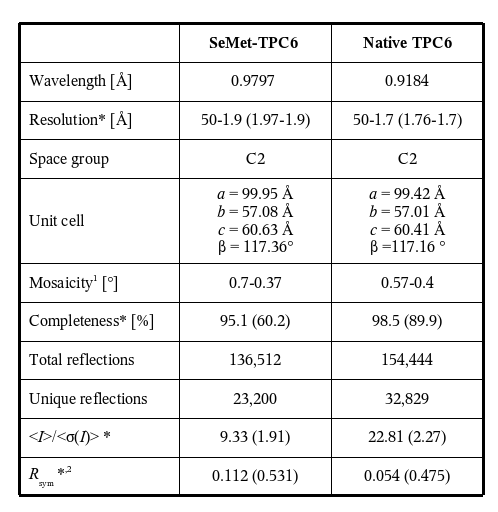
The TRAPP (transport protein particle)-complexes are tethering complexes that play an important role at different trafficking steps of vesicle transport in the recruitment of vesicles to the Golgi network. Two TRAPP complexes have been identified: TRAPP I binds ER (endoplasmic reticulum)-derived vesicles to the Golgi whereas TRAPP II is involved in trafficking within the Golgi. The crystal structure of the TRAPP subunit TPC6 has been determined to 1.7-Angstrom resolution at BESSY Protein Structure Factory beamlines (Table 1), revealing an α/β-plait topology and dimer formation (Fig. 1A, Kümmel et al., 2005). In spite of low sequence similarity, the structure of TPC6 resembles strikingly that of BET3, a further TRAPP subunit, which has also been crystallized as a homodimer (Fig. 1B, Turnbull et al., 2005). The dimerization interfaces of TPC6 and BET3 are also similar, suggesting heterodimerization of both proteins. This was demonstrated by in vivo and in vitro association studies. The TPC6-BET3 interaction could be shown by co-immunopreciptation of epitope-tagged BET3 and TPC6 transiently expressed in HEK 293 cells (Fig. 2A). Crosslinking experiments with recombinant proteins define a heterodimeric stoichiometry of the TPC6-BET3 interaction (Fig. 2B). These data lead to a model of a putative TRAPP subcomplex (Fig. 2C). Together with the TRAPP subunit TPC5, TPC6 and BET3 are hypothesized to constitute a family of paralogous proteins with closely similar three-dimensional structure but little sequence similarity among its members.
Reference
Kümmel, D., Müller, J. J., Roske, Y., Misselwitz, R., Büssow, K. & Heinemann, U. (2005). The structure of the TRAPP subunit TPC6 suggests a model for a TRAPP subcomplex. EMBO Rep. 6, 787-793.
Turnbull, A. P., Kümmel, D., Prinz, B., Holz, C., Schultchen, J., Lang, C., Niesen, F. H., Hofmann, K. P., Delbrück, H., Behlke, J., Müller, E.-C., Jarosch, E., Sommer, T. & Heinemann, U. (2005). Structure of palmitoylated BET3: Insights into TRAPP complex assembly and membrane localization. EMBO J. 24, 875-884.