Department Structure and Dynamics of Energy Materials
Hybrid perovskites
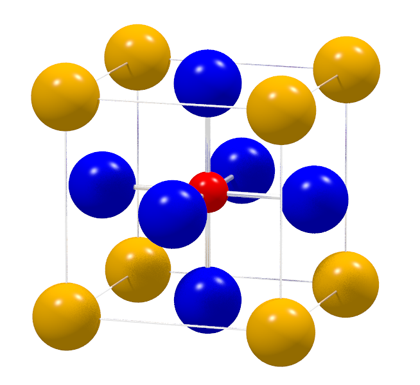
Perovskite aristotype structure: cubic unit cell with A-cation (yellow), BX6-octahedra (B-cation - red and X-anion - blue)
Perovskites with ABX3 – structure show a huge possibility on element substitutions on A-,B- and C-site which leads to a broad variety of physical properties. In recent years the interest become focused on hybrid perovskites as a future photovoltaic material.
Our field of interest lays in lead methylammonium triodide in which A is the organic unit [CH3NH3]+ , B=Pb2+ and X=I3-. The different perovskite crystal structures can be classified by their octahedral coordinated B-cation. The aristotype-structure (P m-3m) is symmetry lowered due to tilting, distortion of [BX6]- octahedra or displacement of B-cation from center of octahedron.
First structure analysis and Rietveld refinements confirmed the MAPbI3 - perovskite belongs to the tetragonal space group I 4/mcm. Further investigations showed that MAPbI3 shows a highly disordered structure with large atomic displacement factors of the anions meaning, that the [PbI6]-octahedra show a tilting. The analyses furthermore revealed a distortion and statistical distributed tilting of the octahedra. This in turn takes influence on the organic unit and therefore influences photovoltaic properties.
For structural investigations Synchrotron and Neutron diffraction experiments are performed at the Helmholtz-Centre Berlin for Materials and Energy.

Tetragonal unit cell of CH3NH3PbI3: light grey - methylammonium molecule on A-site, BX6-octahedra with Pb (dark grey) and I (purple)
