Joint Research Group Catalysis for Energy
Near ambient pressure XPS
Many examples show that the understanding of the interaction of a catalyst´s surface with the reactants plays a key role in a detailed description of catalytic processes. Surface science offers methods for the characterization of almost everything catalysis research could be interested in: Surface composition, morphology, atomic structure, electronic structure, energetics and kinetics of adsorption and desorption. Unfortunately, almost all methods require low pressures and can conventionally not be applied in-situ under the pressure conditions of a realistic reaction experiment. Hence, a spectroscopic characterization of the reacting surface under ambient condition is challenging. The FHI in collaboration with the group of M. Salmeron (Berkley) developed a set-up suitable to record photoelectron spectra in the presence of a reacting gas in the year 2002. An upgraded version is now under supervision of the Energy Catalysis Group (E-GKAT).
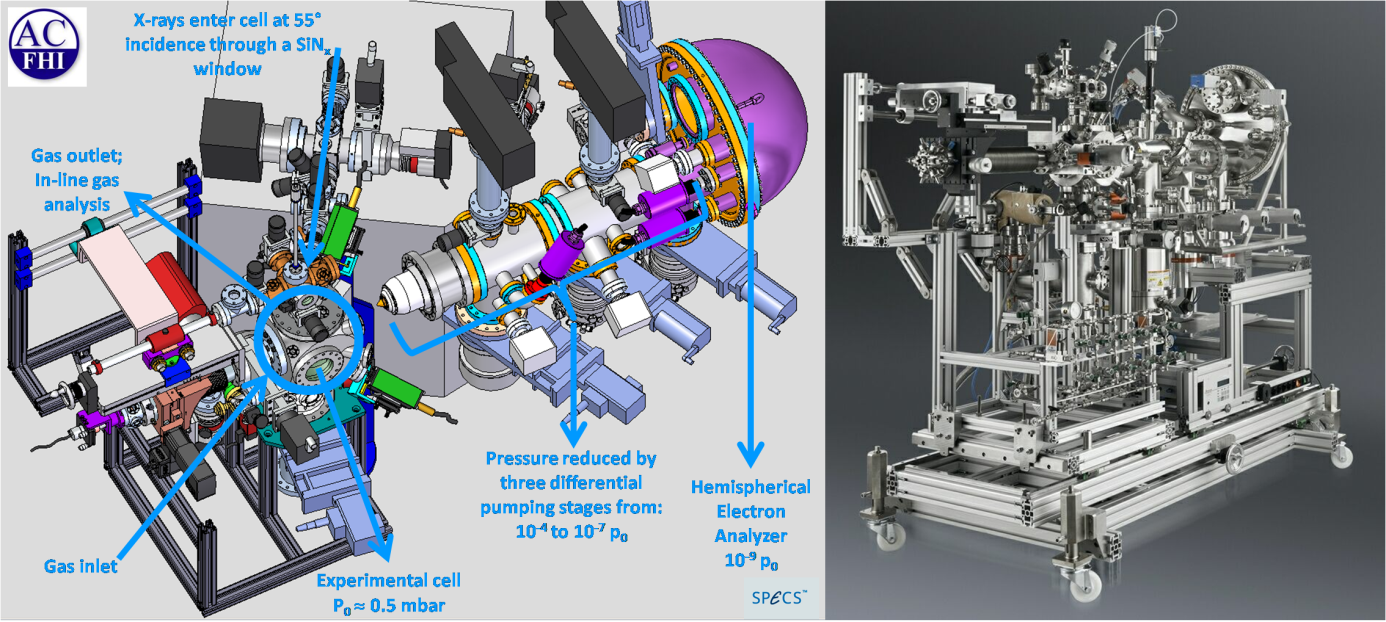
While X-ray photoelectron spectroscopy is a well established technique for surface characterization in high vacuum its application in a gaseous environment is hindered by the strong scattering of the released photoelectrons in the gas phase. The near ambient pressure setup minimizes the signal loss by locating the sample in the vicinity of a differentially pumped aperture system that minimizes the path length of the photoelectron in the gas (Fig. 2).
Simultaneously, the differentially pumped aperture system serves as an electrostatic lens system that increases significantly the acceptance and thus the efficiency of electron collection (compare to Fig. 3a and 3b). Furthermore, this instrument is suitable to collect X-ray absorption spectra (XAS) in the total electron yield mode (TEY) and Auger electron yield mode (AEY). In-line gas analysis can be performed by means of standard mass spectrometry (MS), Proton Transfer Reaction Mass Spectrometry (PTRMS) and Gas Chromatography (GC).
The near ambient pressure XPS set-up is operated with a tuneable X-ray source at the ISISS (Innovative Station for In Situ Spectroscopy) dipole beamline which allows additional modes of operation for in depth analysis and spectral resolution.
Examples for application of near ambient pressure XP spectroscopy can be found in the project section. A general overview on NAP-XPS methodology is given in e.g. D.E. Starr, Z. Liu, M. Hävecker, A. Knop-Gericke and H. Bluhm, Chem. Soc. Rev, 2013, 42, 5833.
High pressure XAS
As an extension to the Near Ambient XPS setup, a High Pressure Soft X-ray Absorption (HP-SXAS) experiment has been constructed and put into operation at the ISISS beamline. This setup allows to measure surface sensitive X-ray absorption spectra of catalysts in the conversion electron yield mode in a broad pressure range between 10 1000 mbar, i.e. up to ambient pressure, at temperatures up to 400°C.
A sketch of the set-up is shown in Fig. 4. Similar to the NAP-XPS, the HP-SXAS setup is rigged in a movable frame which provides the possibility to exchange both machines to be used as endstations at the ISISS beamline.
| Beamline | Contact | |
|---|---|---|
| BElChem-PGM | Berlin Joint Lab for Electrochemical Interfaces @U49-2 PGM | Michael Hävecker |
| ISISS | ISISS | Michael Hävecker |
| Instrument | Contact | |
|---|---|---|
| CAT | CAT@EMIL | Michael Hävecker |
| ECCOS | Energy Conversion Catalysis by Operando Spectroscopy /BElChem-PGM | Michael Hävecker |
| ISISS | Innovative Station for In Situ Spectroscopy | Michael Hävecker |